How Does Ectolysin P128 Work?
Ectolysin P128, GangaGen’s lead ectolysin is a recombinant, chimeric protein that comprises of two functionally distinct domains. The catalytic domain is a muralytic enzyme and the binding domain specifically interacts with the pentaglycine bridge connecting the D-Ala-D-Ala moieties of adjacent peptidoglycan strands. Functionally, P128 is an endopeptidase and its enzymatic action on the staphylococcal peptidoglycan results in the cleavage of the specifc peptide linkages. This leads to the degradation of the cell wall, followed by osmotic lysis and rapid death of the bacterium. P128 targets both coagulase positive and coagulase negative Staphylococci.
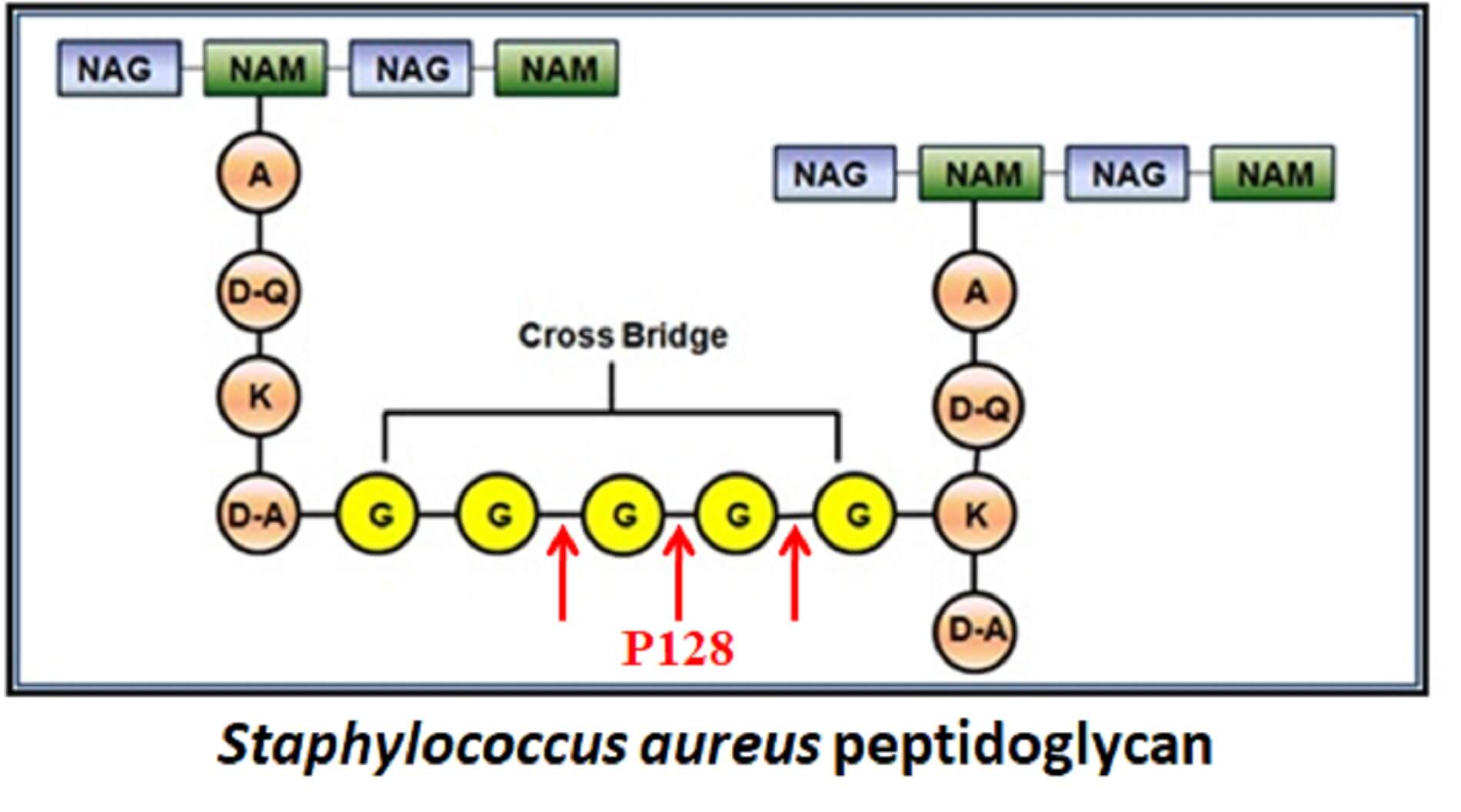
Mechanism of Action
P128 has a novel mechanism of action and has been proven to be very effective both in vitro & in vivo against multidrug resistant pathogens like Methicillin Resistant Staphylococcus aureus (MRSA), Vancomycin Resistant Staphylococcus aureus (VRSA) and Daptomycin Resistant Staphylococcus aureus (DRSA).
Scalable Manufacturing Process
GangaGen has successfully developed the manufacturing process for large scale production of P128 to meet the regulatory standards for clinical studies. P128 formulated as a gel for topical use has been tested in a first-in-human clinical trial, a Phase I/IIA study directed towards treatment of nasal carriage of S. aureus.
P128 Clinical Trial
GBPL advanced its candidate compound into human clinical trials for the decolonization of Staphylococcus aureus from human nares.
Intravenous Treatment
GBPL is also advancing Ectolysin P128 for intravenous treatment of bacteremia caused by Staphylococcus Species.